Ophioglossum vulgatum | Adder’s Tongue Plant (2024 Guide)
Table of Contents
Ophioglossum vulgatum also known as Adder’s Tongue Plant belongs to the family order Ophioglossaceae. The order contains this single family. There are two orders in the class Eusporangiopsida.
These orders are Ophioglossales and Marattiales. The class Eusporangiopsida is characterized by the following features.
The sporangium in all pteridophytes is initiated by the laying down of a cross-wall in a superficial cell or a group of cells. Since this wall is periclinal each initial cell is divided into an outer and inner daughter cell.
If the sporogenous tissue is derived from the inner daughter cell, the sporangium is described as ‘eusporangiate’ and if from the outer, as ‘leptosporangiate’. Here each sporangium originates from a group of initial cells.
The wall of sporangium consists of more than one layer of cells. The sporangium contains a very large number of spores. The antheridia remain sunken in the gametophytic tissue and usually contains many antherozoids.
Ophioglossum vulgatum (Adder’s Tongue Plant) Classification
The characteristic features of the order Ophioglossales are as follows:
Characteristics Of Order Ophioglossales
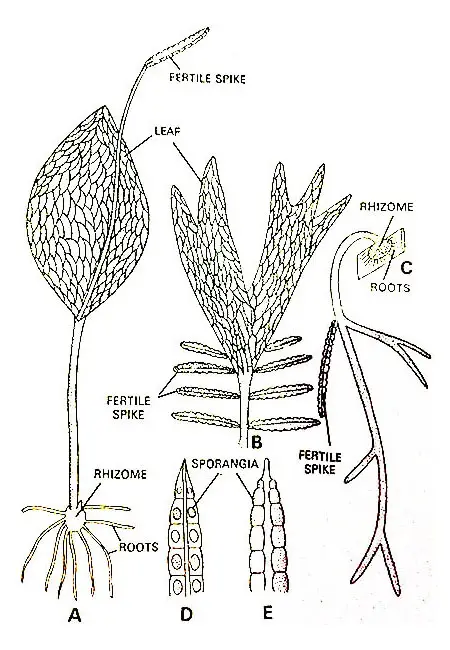
The order is represented by herbaceous sporophytic plants. The sporophyte possesses a short, fleshy, naked rhizome that is erect or dorsiventral.
The sporangia are borne on an outgrowth, the fertile spike which projects from the adaxial surface of the leaf usually near the juncture of the blade and petiole.
The sporangia are found on the margins of the fertile spikes. The number of spores in each sporangium is very large.
The spores are of one kind, i.e., homosporous. The prothalli developed from the spores are saprophytic, tuberous, and subterranean.
Classification Of Ophioglossum
All the details about genus along with order have been discussed here in detail.
- Systematic Position: Pteridophyta
- Division: Pterophyta
- Class: Eusporangiopsida
- Order: Ophioglossales
- Family: Ophioglossaceae
- Genus: Ophioglossum
Distribution And Habitat
According to Clasuen (1938), there are 8 species in this genus. They are worldwide in distribution. Many species are found in temperate regions, but this has least thrived in the tropics.
About a dozen species are found in our country. The species are Ophioglossum vulgatum, Ophioglossum pendulum, Ophioglossum nudicaule, Ophioglossum costatum, Ophioglossum lusitanicum, and Ophioglossum aitchisoni.
Most of the species of Ophioglossum are terrestrial plants and commonly grow in humus soil. Two species are epiphytic, e.g., O. pendulum also called ribbon fern plant, and O. palmatum. These species grow on trunks and branches of trees.
This genus has been divided into three sub-genera 1. Euophioglossum; 2. Ophioderma and 3. Cheiroglossa.
External Morphology Of Ophioglossum
Sporophyte
The sporophyte of most species of Ophioglossum possesses a short, erect, subterranean rhizome. A few species (e.g., O. pendulum; O. intermedium) have a markedly dorsiventral stem.
The branching of the sporophyte is generally due to the formation of axillary buds in the axils of leaves. Petry (1915) has also described the branching of rhizomes as dichotomous.
Root
The rhizome bears numerous Adventitious roots. These roots are commonly thick, smooth, and devoid of hairs.
As a rule, the roots are borne singly below the leaf or leaves of the current season and the leaf scars of previous seasons. The roots are mycorrhizal. They arise endogenously.
Leaves
Most of the species of Ophioglossum produce but one leaf each year. Some tropical species may produce four or five leaves in a season. The leaves are usually borne in an irregular spiral on the stem.
Where growth is seasonal the development of the leaf is very slow. In and cases the embryonic leaf at a stem apex develops slowly for three years and then grows about ground and unfolds the fourth year.
A fully expanded leaf is sharply differentiated into blade and petiole. The blade of the leaf in all but one species (O. palmatum) is entire and narrowly linear to broadly oval in outline.
At the juncture of the blade and petiole, in all but one species (O. palmatum) there is a single fertile spike. In O. palmatum there are several small fertile spikes in the region where the blade and petiole adjoin.
In O. palmatum the blade is palmately incised into many narrow segments. In O. pendulum the leaf may attain a length of 1.5 meters or more and its pedicle merges gradually into a strap-shaped lamina.
The venation of the blade is always reticulate. There is no distinct midrib, but according to the species the small veinlets may or may not be present. These veinlets end blindly within the large meshes.
However, the veins near the central axis of the blade are more conspicuous than the remaining veins. The venation of the leaf is erect and does not circinate.
Apical Growth
A single inconspicuous pyramid-like apical cell with three cutting faces is found to be situated at the apex of the stem.
This apical cell cuts off derivatives that divide and re-divide forming an embryonic tissue that becomes differentiated into stele, cortex, and epidermis on its maturity.
Anatomy (Internal Morphology)
Anatomy Of Rhizome
A transverse section of the rhizome reveals an irregular outline due to the attachment of Adventitious roots and leaf bases.
The cortex remains bounded by a single-layered epidermis. The cortex consists of large thin-walled parenchymatous cells having abundant intercellular spaces among them.
Maheshwari and Singh (1934) reported the formation of a periderm layer in old rhizomes, but there is no functional cambium.
The lowermost portion of the rhizome may have an endodermis delimiting the stele and cortex (Bower, 1911). In the upper portion of the rhizome, there is no endodermis.
Here the innermost cells of the cortex and the outermost cells of the steles are so similar in appearance that it becomes difficult to differentiate where one region leaves off and begins.
Lang (1912) has reported internal and external endodermis in young plants of the Ceylonese species of Ophioglossum.
Generally, the rhizome possesses an ectophloic siphonostele throughout its entire length. This siphonostele may have but one leaf gap when cut at any particular level.
More often two or more leaf gaps may overlap forming a dictyostelic condition. Most of the species have a single trace departing at each gap and traversing the cortex without branching.
A few species. e.g. O. palmatum and O. pendulum have two traces departing at each gap. There is a division of each trace into several strands before it runs into the leaf base.
The xylem of each meristele is endarch in arrangement. The protoxylem remains situated on the inner face of the xylem mass. The reticulate tracheids of irregular shape form the metaxylem.
Boodle (1899) has reported a small amount of secondary thickening in the stem of O. vulgatum. The protoxylem is composed of spiral and scalariform elements.
In O. palmatum, the protoxylem is ill-defined. The relation of the xylem to the phloem is collateral, that is the phloem lies external to the xylem.
Commonly the phloem consists of a layer of four or five cells in thickness. However, in O, pendulum (Petry, 1914) the phloem consists of a single layer of cells separated from the xylem by a layer of parenchyma three to five cells in thickness.
The pith internal to the xylem is composed of thin-walled large parenchymatous cells. According to Petry (1914), sometimes (e.g., in O. pendulum) the tracheids occur in the pith region.
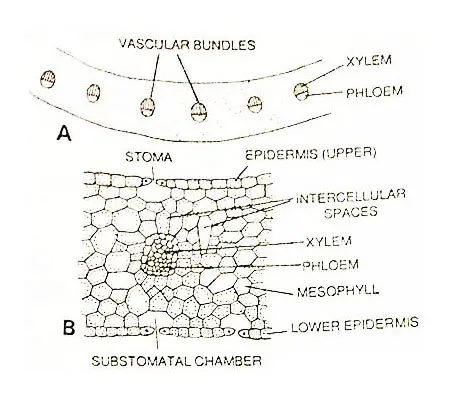
Anatomy Of Leaf
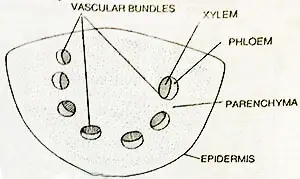
The epidermis of the petiole is single-layered. The interior of a petiole is composed of spongy parenchyma that may or may not possess conspicuous cavities in the central region.
Just beneath the epidermis, the parenchymatous cells contain numerous discoid chloroplasts.
The number of vascular bundles is one or two at the base. The trace or the pair of traces departing to a leaf becomes divided into strands that lie parallel to one another throughout the length of the petiole.
The bundles may be arranged in a crescent or c-shaped outline. The bundles are collateral. The xylem is endarch in development. The phloem is found toward the outer surface.
The internal structure of the sterile leaf lamina resembles that of the petiole. In the species with erect leaves, stomata are present in equal numbers on both faces of the blade.
In O. reticulatum, where the leaf blade is more or less horizontal, the upper epidermis contains more stomata. There is no distinct hypodermis.
The bulk of lamina is composed of thin-walled green cells with frequent intercellular spaces. The structure is similar to that of a petiole.
Several veinlets are found to be present in the leaf lamina. All cells of mesophyll tissue contain chloroplasts.
Anatomy Of Root
A transverse section of the root reveals a single-layered epidermis, a broad cortical region, and a central stele. In the young root, the epidermis remains intact and the root hairs are lacking.
In the older roots, the epidermal cells are thickened and suberized. According to Maheshwari and Singh (1934), in O. fibrosum the epidermal layer is soon replaced by an exodermis developed from the outer cortex:
The cortex is sufficiently broad and has the innermost cells differentiated into an endodermis with well-defined Casparian strips. The rest of the cortical region consists of two concentric regions of approximately equal thickness.
The outer cortex is 4 to 7 cells in thickness and is composed of angular cells without intercellular spaces. A mycorrhizal fungus is also present in the outer cortex. This is a phycomycetous fungus.
The inner cortex is composed of rounded cells with intercellular spaces. These cells frequently contain starch grains.
The stele of the root is generally monarch or diarch but tri-tetra and even Petrarch steles have been observed in different species of Ophioglossum.
In the diarch roots, the usual plate of xylem possesses a protoxylem mass at each pole. There is a mass of phloem on either side of the xylem.
Each of the xylem masses is separated from the phloem by a parenchymatous tissue two to three cells in thickness.
In certain monarch roots, the xylem and phloem are to be found radial to each other. In other cases, the xylem and phloem masses lie with their broader sides facing each other.
Vegetative Reproduction
The vegetative propagation takes place by the adventitious buds formed on the roots as found in O. pendulum and O. aitchisoni.
Spore Producing Organs (Sporangia)
The sporangia are borne on fertile spikes. The fertile spikes arise from the basal portion of the sterile leaf. The fertile spike may easily be differentiated from the sterile segment. It is usually erect and stalked.
The sporangia are found within the tissue of the fertile spike. The sporangia are found to be arranged on the margins of the spike in two longitudinal rows. The number of sporangia varies from 6 to 20 on a fertile spike.
Development Of The Spike
A pyramid-like apical cell with four cutting faces gives rise to a conical structure of meristematic tissue that represents a very young fertile spike. This conical outgrowth turns upwards.
According to Bower (1896), the four quadrants cut just back of the apex of the spike corresponding to the four cutting faces of the apical cell.
These quadrants are oriented in such a way that two lie in a plane perpendicular to the sterile lamina, and two in a plane parallel to it.
Development Of Sporangium
As the development of the spike continues and it becomes older, a vertical strip of cells in the epidermal layer of the two quadrants parallel to the leaf blade becomes differentiated.
Each such strip two to three cells broad and several cells tall is a sporangiogenic band. The cells of the sporangiogenic band divide periclinally and anticlinally forming the two bands that become three or four cells in thickness.
Now the hypodermal region of the sporangiogenic band differentiates into alternate blocks of sterile cells and archesporial cells.
Each group of archesporial cells divides repeatedly forming a large number of sporogenous cells. The portion of the band external to each archesporium divides periclinally to form a multilayered jacket layer of the sporangium.
The sterile cells also divide vertically again and separating the sporangia one from another. As the sporogenous tissue continues development, it becomes surrounded by a poorly developed tapetal layer.
According to Bower (1908), the tapetum has been developed from outermost sporogenous cells. But it seems more probable that the tapetum arises from cells found immediately external to the sporogenous mass.
The tapetum consists of several poorly developed layers of cells that ultimately, break down into a plasmodial mass with persistent free nuclei.
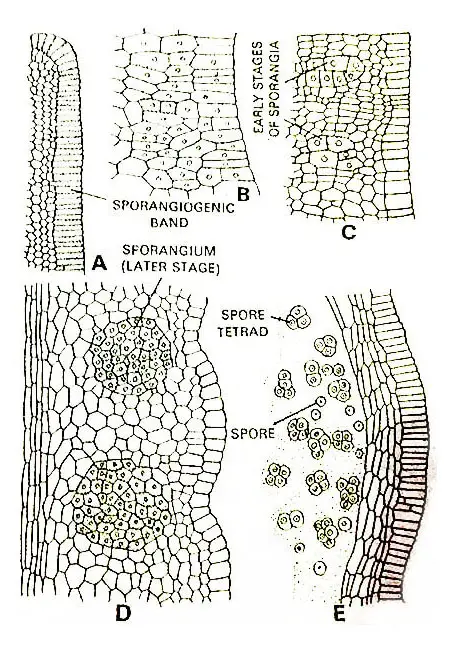
This plasmodial mass surrounds the isolated spore mother cells and supplies nourishment to them.
The spore mother cells divide meiotically forming the tetrads of haploid spores. The mature sporangia remain embedded within the tissues of the spike.
Each sporangium contains a very large number of spores, sometimes this number reaches up to 10,000.
Dehiscence Of The Sporogonium
Each sporangium of the fertile spike splits by a transverse slit on the maturity of the spores. However, there is no special mechanism through the annular causing the opening of the sporangial wall.
Dehiscence takes place by drying out and shrinking the sterile tissues within the spike.
Morphological Nature Of Fertile Spike
The morphological nature of the fertile spike is controversial. According to Bower (1896, 1908), it is a single septate sporangium but this hypothesis has been rejected later on by bower (1911, 1926) himself.
The pteridologists of modem days agree that in nature it is pinna-like. It has been held by Goebel (1915) that the spike represents a single pinna.
But according to Chrysler (1910), it is more probable that the fertile spike represents the fusion of the two basal pinnae of a leaf.
This interpretation has been supported based on vascular supply. The fertile spike has the same vascular supply as would a pair of pinnae.
Gametophyte
Spores
The spores are of tetrahedral type. The spore wall consists of two layers, the exospore, and the endospore. The exospore is thick, colorless, and sculptured with pits.
The endospore is thin and delicate. The spore contains granular cytoplasm, a centrally placed large nucleus, and oil droplets.
Germination Of Spore and Development Of Prothallus (Gametophyte)
Germination of spores to form gametophytes takes place immediately after shedding.
Sometimes the germination of spore is greatly delayed (Campbell, 1911).
On absorption of water, the exospore ruptures along the triradiate ridge, and the inner contents of the spore surrounded by the endospore come out in the form of a germinal tube.
This divides transversely into two cells. The next division is vertical in the lower cell and divides it into two equal cells. At this stage, the young gametophyte consists of three cells.
The young gametophyte does not develop beyond the three or four-celled stage unless it becomes infected with the phycomycetous fungus.
The fungus always enters through the lower cell and establishes as a mycorrhizal endophyte.
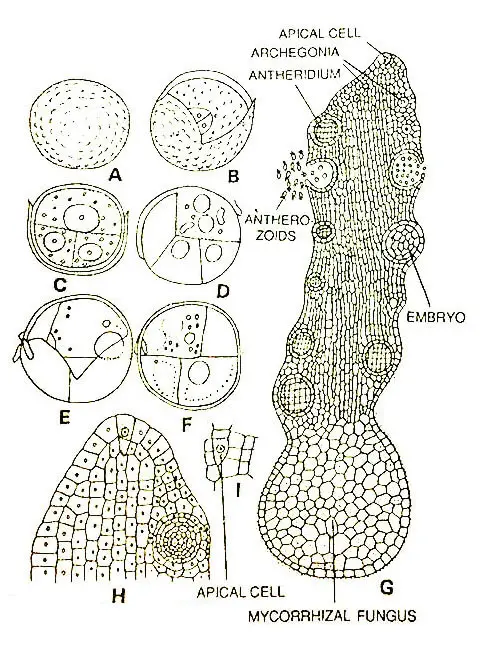
Mature Prothallus
The mature gametophytes (Bruchmann, 1904; Campbell, 1907; Lang, 1902) are irregularly cylindrical to conical.
According to the species, the gametophytes may be unbranched (e g. O, vulgatum) or profusely branched (e.g., O. pendulum) the gametophyte is a subterranean inhabit and obtain all its food through the agency of a mycorrhizal fungus.
Sometimes it develops small green lobes above ground. The size of the prothallus ranges from 5 to 10 cm.
In most species the gametophyte is perennial, but in O. moluccanum the prothalli seem to be annual (Campbell. 1911). The growing apex of a prothallus has a single apical cell with three or four cutting faces.
The prothalli are usually pale, grey, yellowish, or brownish. However, the region immediately posterior to the apical cell is colorless.
According to species the prothalli may or may not bear rhizoids. Internally there is little differentiation of tissue within a gametophyte, but the cells towards the interior may be somewhat elongated.
Interior cells in the older parts of a prothallus are infected with a mycorrhizal fungus. However, this endophytic fungus is not found in the apical portion of the thallus and the outer cells of the older portion of the thallus.
The Sex Organs
The prothalli are monoecious and bear sex organs in acropetal succession. The antheridia and archegonia are found to be developed in irregular sequence. These sex organs remain scattered all over the prothallus.
Development Of Antheridium
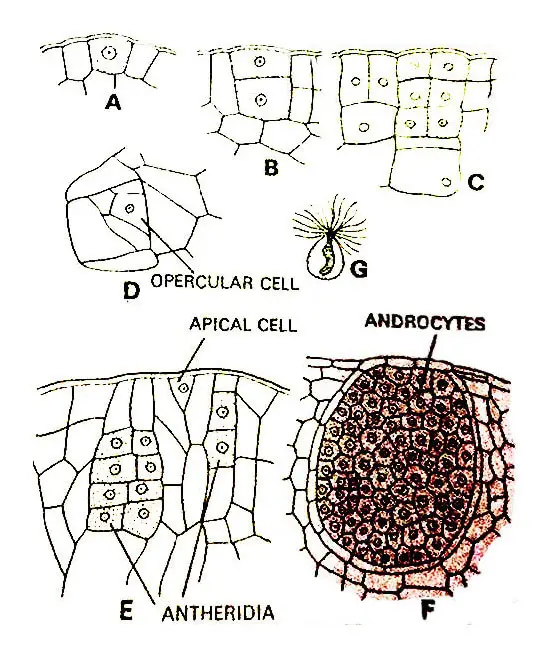
The antheridium develops from a single superficial cell of the prothallus.
This antheridial initial divides periclinally to form an outer cell, the jacket initial, and an inner cell, the primary androgonial cell.
According to Campbell the Jacket initial always divides anticlinally to form two daughter cells of equal size.
One of these daughter cells again divides anticlinally forming two daughter cells of which one is approximately triangular and the other approximately rectangular.
The triangular cell functions in the same manner as an apical cell and cuts off a series of cells from each of its three sides.
After it has stopped cutting off cells the triangular cell itself is known as an opercular cell.
The primary androgonial cell divides anticlinally or to form two androgonial cells. The next division takes place in the opposite plane and this way the four androgonial cells are found to be arranged in two tiers.
These four androgonial cells divide and redivide continuously to form numerous (several thousand) androgonial cells. Members of the last cell generation of androgonial cells are known as Androcyte Mother Cells.
Androcytes metamorphose into antherozoids. Each androcyte has two blepharoplasts at one side of the nucleus. Campbell (1911) has studied metamorphosis.
The first noticeable change in the metamorphosis is an elongation of the blepharoplast. Thereafter the nucleus elongates slightly.
As metamorphosis proceeds the blepharoplast becomes coiled and the nucleus also becomes coiled. Shortly after this several flagella develop at the anterior portion of the blepharoplast.
The mature antheridia are usually large and sunken within the tissue of the prothallus. Sometimes they are slightly projecting. The jack layer of antheridium is single-layered.
On being mature of the antheridium the opercular cell becomes brownish and disintegrates. Eventually, the antherozoids liberate through this triangular pore.
The mature antherozoids are coiled and multi-flagellate. The narrowly coiled anterior portion of the antherozoid consists mainly of cytoplasm and blepharoplast. The broad, loosely coiled posterior portion is mainly the nucleus.
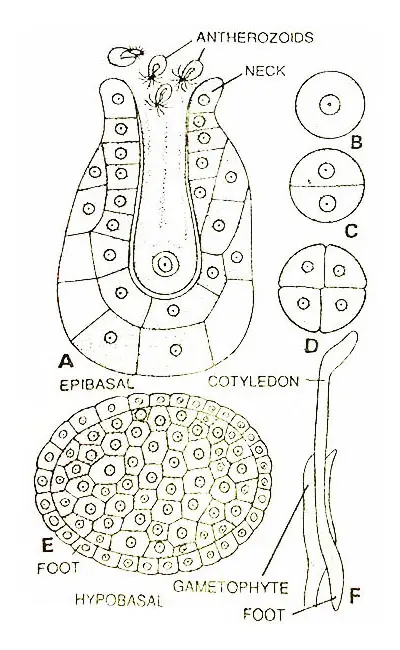
Development Of Archegonium
The archegonium develops from a single superficial cell found to be situated near the apex of the prothallus. This cell is archegonial initial.
The initial divide periclinally to form a primary cover cell and an inner cell. Now the inner cell divides periclinally to form a central cell and a basal cell. The central cell lies next to the primary cover cell.
Thereafter the primary cover cell divides anticlinally to form two daughter cells that divide anticlinally forming the four quadrately arranged neck initials.
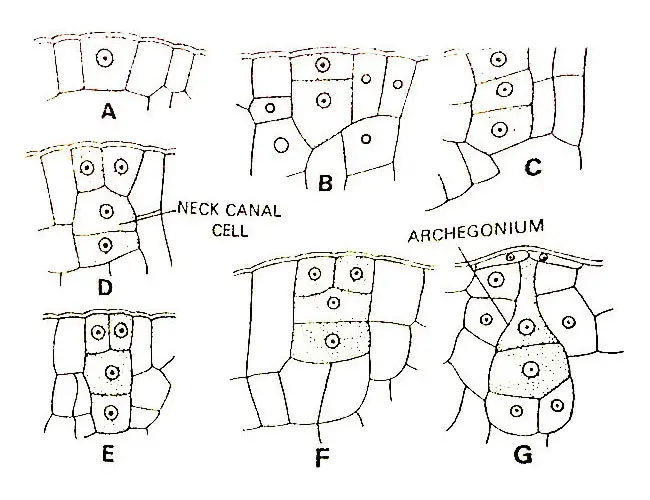
Now each of the neck initials divides diagonally by a transverse division resulting in the formation of a neck composed of two tiers of four cells each.
Later more transverse divisions take place in each tier and a mature neck with three or four cells in height is formed.
As the neck develops the central cell divides periclinally to form a primary canal cell and a primary venter cell. The primary canal cell elongates vertically and pushes out into the neck.
The large and conspicuous nucleus of the primary canal cell divides to form two canal cells. Shortly before the maturation of the archegonium, the primary venter canal cell undergoes a division forming a venter canal cell and an egg.
The venter canal cell immediately disintegrates after its formation. The venter canal cell is a transitory structure and it is rarely demonstrable (Campbell, 1907).
A nearly mature archegonium consists of a neck composed of four vertical rows of cells and it is three to four cells in height. The archegonium remains embedded within the tissue of the prothallus or it projects out very slightly.
On the maturation of the archegonium, the cells disintegrate and a canal is formed. One of the multi-flagellate antherozoids enters through the neck canal and fuses with the egg thus forming the diploid oospore.
Development Of Sporogonium
The zygote divides transversely to form an outer epibasal and inner hypobasal cell.
According to Campbell (1907), the hypobasal cell develops only into the foot while according to Bruchmann (1904) it gives rise to both the foot and primary root. However, an apical cell becomes differentiated in the root portion of an embryo.
According to Bruchmann (1904) in O. vulgatum development of root and foot so over masks that of cotyledon and stem that the young sporophyte appears to be all root for a long time after it comes out from the prothallus.
According to Campbell, in O. moluccanum, however, the root and the cotyledon grow at an equal rate. The root grows downwards while the cotyledon upwards.
The method of formation of the cotyledon from the epibasal cell is not known. However, the cotyledon develops through an apical cell.
After the earlier development of the primary root and cotyledon, there appears an apical cell of the stem. This appears in the region where cotyledon and root adjoin.
This apical cell seems to be a superficial cell of the embryo. The stem from this apical develops very slowly.
According to Campbell (1907), in O. moluccanum the development of an embryo, an independent sporophyte with cotyledon and primary root may take one year. It may take several years in the case of O. vulgatum.
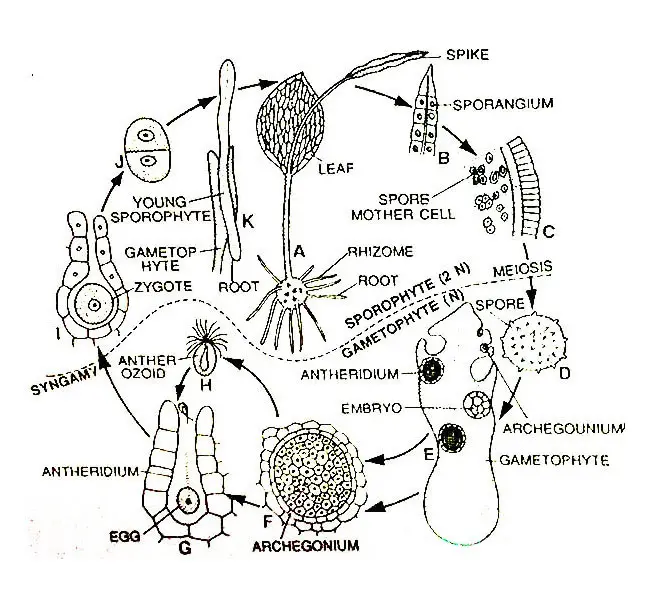
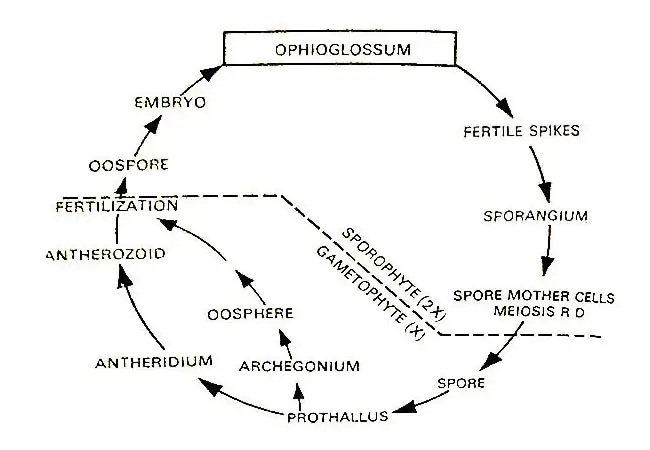
Life Cycle Of Ophioglossum
Below is the detailed graphical and diagrammatic life cycle of Ophioglossum.






I think this is among the most vital info for me. And i’m satisfied studying your article. However should commentary on few normal things, The website taste is ideal, the articles is in reality excellent. Excellent activity, cheers.