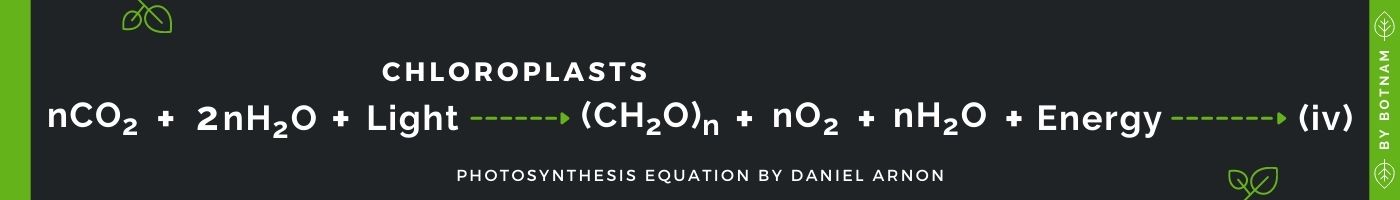Photosynthesis Definition & History of Photosynthesis 2024
Table of Contents
Photosynthesis definition in terms of science is “the conversion of light energy into useful chemical energy”.
Martin Kamen’s (1963) photosynthesis definition is described as the series of processes in which electromagnetic energy is converted to chemical-free energy that can be used for biosynthesis.
The more comprehensive photosynthesis definition could be “The process in which certain carbohydrates are synthesized from carbon dioxide and water in the chlorophyll cells in the presence of sunlight and oxygen is being liberated in the process.”
Photosynthesis can be singled out as a phenomenon, which is responsible for life on the earth. For animals and plants alike, it is the only source, which provides energy (Food) to all.
For animals besides food oxygen is also provided through this process. This process remains a mystery till many scientists involved with photosynthesis discovery came up with definite results.
A sum-up of all the efforts contributed by all these workers made us understand this phenomenon, as we know it now.
History of Photosynthesis
The disclosure of the mechanism involved in Photosynthesis is extended over three centuries.
The landmarks are illustrated to follow the footsteps of those who make us understand one of the basic mechanisms involved in the maintenance of life on earth.
Stephen Hales
In 1727, Stephen Hales took the first step when suggesting that part of plant nourishment came from the atmosphere and light participates in this process.
Before him, scientists believed that plants obtained all of their required elements from the soil.
Joseph Priestly
In 1771, Joseph Priestly discovered O2 and found that green plants could renew air which is made “bad” by the breathing of animals. He performed the famous experiment of mice mint and candle.
Jan Ingenhousz
In 1779, Jan Ingenhousz a Dutch physician demonstrated that light was necessary for this purification of air. He found that plants, too, made “bad air” in darkness.
Jean Senebier
In 1782, Jean Senebier showed that the presence of the “bad” gas produced by animals and by plants in darkness (CO2) stimulated the production of “purified air” (O2) in light.
So, by this time the participation of two gases in photosynthesis had been demonstrated. Lavoisier and others made it apparent that these gases were CO2 and O2.
T De Saussure
In 1804, N T de Saussure associated water with the phenomenon and made the first quantitative measurements of photosynthesis.
He found that plants gained more dry weight during photosynthesis than could be accounted for by measuring CO2 absorbed to the weight of O2 released.
He attributed the difference to the uptake of H2O. He also noted that approximately equal volumes of CO2 and O2 were exchanged during photosynthesis.
Julius Sachs
In 1864, Julius Sachs demonstrated the production of other chemical products of photosynthesis-organic matter.
He observed the growth of starch grains in illuminated chloroplasts. Starch was detected only in areas of the leaf exposed to light.
The photosynthesis equation was demonstrated to be as follows:
C B Van Niel
In the 1930’s C B van Niel made an important discovery and pointed out the similarity between the overall photosynthetic process in green plants and also in certain bacteria.
Various bacteria were known to reduce CO2 using light energy and an electron source other than water. (Some of these bacteria use organic acids such as acetic or succinic acid as electron sources).
Whereas those to which van Niel gave primary attention use H2S and deposit sulfur as a by-product. The overall photosynthesis equation for these bacteria was believed to be as follows:
When Reaction (i) is compared with Reaction (ii) similarity can be seen between the roles of H2S and H2O, and between those of O2 and sulfur.
This suggested to van Niel that the O2 released by plants is derived from water, not from CO2.
Robin Hill And R Scarisbrick
In the late 1930’s Robin Hill and R Scarisbrick supported this idea. Their work showed that even isolated chloroplasts and chloroplast fragments could release O2 in the light if they were given a suitable acceptor for the electrons being removed from H2O.
Hill Reaction
Certain ferric (Fe3+) salts were the earliest electron acceptors provided to the chloroplasts, and they became reduced to the ferrous (Fe2+) form. This light-driven split (photolysis) of water in the absence of CO2 fixation became known as the Hill Reaction.
Hill and Scarisbrick’s work showed that whole cells were not necessary for at least some of the reactions of photosynthesis and that the light-driven O2 release is not required as tied to the reduction of CO2.
Samuel Ruben And Martin Kamen
In 1941 Samuel Ruben and Martin Kamen gave more convincing evidence that the O2 released derived from H2O.
They supplied the green alga Chlorella with H2O containing 18O, a heavy, non-radioactive oxygen isotope that can be detected with a mass spectrometer.
The O2 released in photosynthesis became labeled with 18O, supporting van Niel’s hypothesis.
Thus, in the summary for the photosynthesis equation given in Reaction (i) to include two H2O molecules as reactants we get:
In 1951, it was found that plants constituent a coenzyme, (nicotinamide adenine dinucleotide phosphate) (NADP+).
It could act as a Hill reagent by accepting electrons from water in reactions occurring in isolated thylakoid membranes or broken chloroplasts.
This discovery again stipulated photosynthesis research; because it is known that the reduced form of NADP+ to NADPH, could transfer electrons to many plant compounds, it was correctly suspected that its normal role in chloroplasts was a reduction of CO2.
Therefore, one of two essential functions of H2O is to reduce NADP+ to NADPH; the other function is to provide energy to form ATP from ADP and Pi.
Daniel Arnon
Photophosphorylation: In 1954 Daniel Arnon found that ATP was synthesized in isolated chloroplasts only in light, and the process was known as photosynthetic phosphorylation, or simply photophosphorylation (addition of phosphate in light).
Oxidative Phosphorylation: Before that ATP formation was restricted to respiration, reactions that occur in the mitochondria are known as oxidative phosphorylation.
This process of ATP formation by photophosphorylation can be summarized as follows:
Photophosphorylation in chloroplasts accounts for much more ATP formation in leaves during daylight than does oxidative phosphorylation in mitochondria.
ADP and Pi are rapidly converted to ATP by light energy, and the ATP is just as rapidly broken down in the process of CO2 reduction and carbohydrate synthesis when photosynthesis is occurring at a constant rate.
The equation can be depicted as:
For Equation iv to proceed to require CO2 and H2O, which combines in the presence of light at the site of chloroplasts as a result carbohydrates (CH2O), Oxygen (O2), and water (H2O) are produced.
Measurement Of Photosynthesis
If we look at equation (iv) we find three points where photosynthesis can be measured. These points are the amount of carbon dioxide absorbed, amount of oxygen evolved, and amount of carbohydrates produced.
The Common methods used for the determination of photosynthesis also depend upon the measurement of these factors.
Net Photosynthesis
All these methods are subjected to errors because the respiration phenomena are also going on side by side. The oxygen evolved during photosynthesis is partially used up in respiration hence the amount of O2 evolved cannot be exact measurements.
Similarly, CO2 evolved during respiration is absorbed during photosynthesis. Therefore, the exact quantity cannot be measured by these methods. The photosynthesis measured in this way is known as apparent or net photosynthesis.
True Photosynthesis
The error because of respiration can be removed by determining the rate of respiration in the dark and then detecting photosynthesis.
The Photosynthesis measured in this way is known as real or true photosynthesis. One objection that remains is that the rate of respiration in dark may differ from the rate in light.