Structure of Water Molecule | Properties of Water 2024
Table of Contents
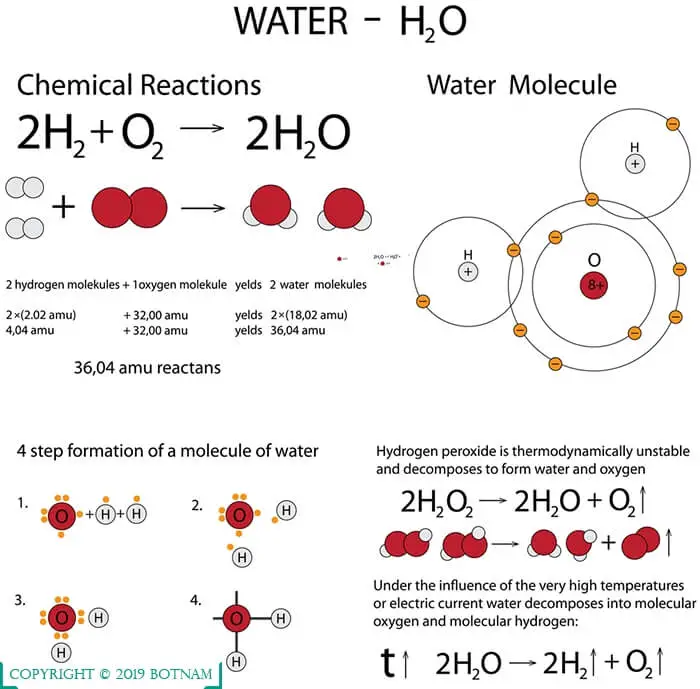
The structure of water molecule is made up of one molecule of oxygen and two molecules of hydrogen-bonded covalently.
Water (H2O) is essentially considered one of the most important substances found on the earth. It covers over 70% of the earth’s surface and makes up as much as 95% of the living organisms.
The Structure of Water Molecule
It is virtually unique among liquids because of its molecular structure. As a result of the strong attraction of the oxygen nucleus for the electrons of hydrogen, the hydrogen atoms are distorted from their usual position.
This results in the formation of an angle (105°) between hydrogen atoms. Because of this asymmetry of hydrogen atoms water has a strong dipole movement, i.e., it is. highly polarized with a strong separation of positive and negative charges.
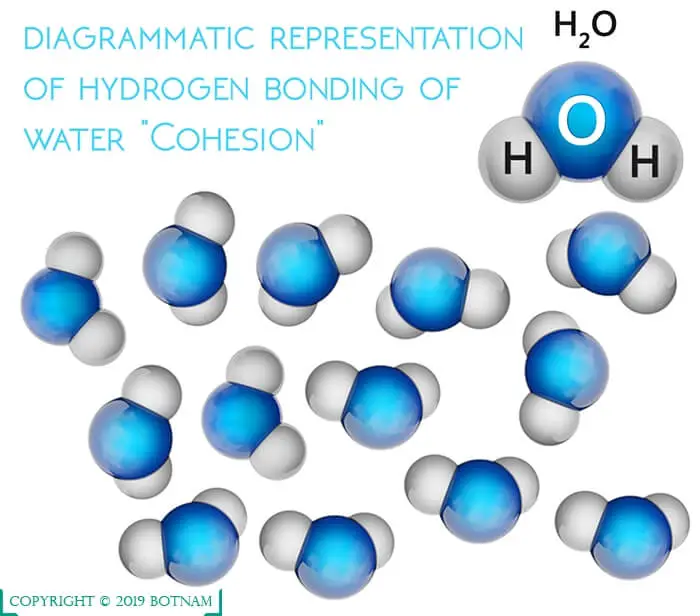
Because of this polarization, it readily shares its positively charged hydrogen with the negatively charged oxygen of other molecules.
Thus, water becomes very cohesive, which results in the development of strong bonding with itself, and adhesive, binding strongly with other molecules containing oxygen. This structure of water molecule executes certain properties in it.
What Are The Properties of Water?
Cohesion in Water
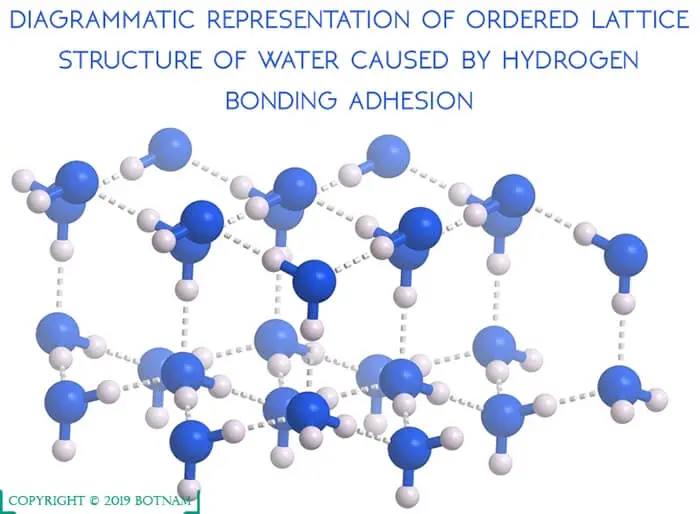
The cohesion in water molecules themselves results in a very ordered structure. When they are frozen as ice, a definite pattern exists.
In liquid form, the pattern is altered, as ice melts, hydrogen bonds are broken and water increases in density up to a temperature of 4°C as the pattern collapses and becomes denser.
When the temperature increases from 4°C more bonds are broken because of thermal agitation, and water becomes less dense. At 100°C hydrogen bonds may break completely, resulting in the escape of water molecules as vapor.
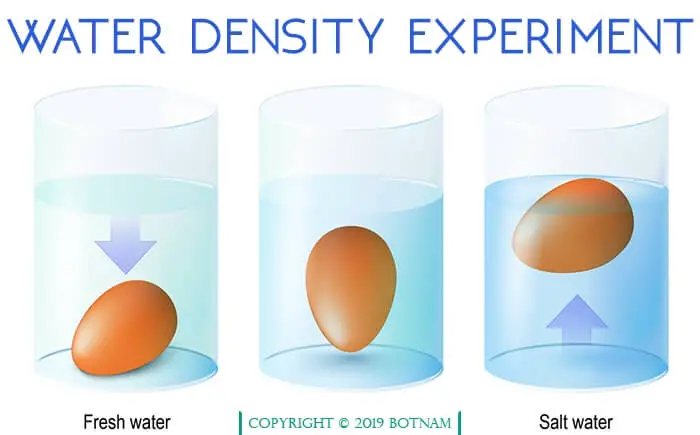
The maximum density of water occurs at 4°C rather than at a minimum temperature like most substances have a profound effect on aquatic life.
Ice at 0°C has a density of 0.917 g cm-3 whereas water at 0°C has a density of about 0.999 g cm-3 and at 4°C is 1.0 g cm-3. Hence water freezes from the top down rather than from the bottom up.
The frozen water at the surface tends to insulate the water below, preventing large bodies of water from freezing solid and killing aquatic life.
Adhesion of Water
The adhesion of water is explained by the hydrogen bonding of water molecules to other polar surfaces.
The strong dipole of water exerts electrostatic and gravitational forces on charged electrovalent compounds and on the dipoles of polar, covalent compounds.
Thus, water will adsorb to substances such as cellulose (cotton) but not to polyesters with little oxygen available for hydrogen bonding.
This asymmetrical structure results in electrostatic attraction for other dipoles and ions and in hydrogen bonding with oxygen-containing molecules.
However, the hydrogen bond is relatively easy to break when compared with a covalent bond since it has bond energy in the range of only 10 to 30 kilo-joules (kJ) per mole but a large number of hydrogen bonds give the gigantic bio-molecule considerable strength.
Heat of Vaporization
The heat of vaporization, the amount of heat required to convert one unit of liquid to vapor, is excessively high for water as compared to other liquids.
The high heat of vaporization of water 2435 J g-1 at 25°C (582 cal g-1) results because of its cohesive nature. Perhaps the most important consequence of water’s high heat of vaporization is the cooling effect as water evaporates from the living surface.
Viscosity of Water
The viscosity of water, or resistance to flow, is higher in water than in most liquids, again because of the hydrogen bonding.
The viscosity of water at 20°C taken as the reference is 1.0 centipoise: Ethanol has a viscosity at 20°C of 1.2 centipoise and hence is slightly more viscous, but ethyl ether has a viscosity of 0.2 centipoise at 20°C.
Temperature effects on the transport of liquid within plants can be partially accounted for by viscosity changes.
Volume and Density
Water is most dense at about 4°C and less dense above and below 4°C. It is at least at 4°C. As temperature increases or decreases from 4°C the volume occupied by one gram of water increases.
Thus, at 0°C water will have a volume of 1.00012 cm3 g-1, and ice will be 1.09 cm3 g-1. Water at 20°C will be 1.00177 cm3 g-1.
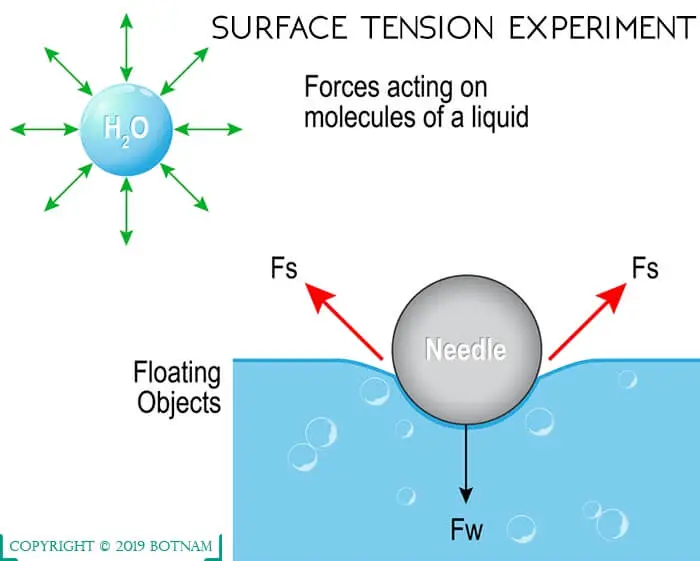
Surface Tension of Water
The tendency of the water surface to contract and resemble an elastic membrane is known as the surface tension of water. Water drops tend to form spheres. In this state, it has less energy per unit of the surface relative to volume.
The energy associated with the surface is surface tension and can be expressed in units of dynes per centimeter (dyn cm-1. As a result of surface, tension liquids rise in the capillary tubes. The rise of the capillary is very important for consideration of the rising of water in trees.
Specific Heat of Water
The specific heat of water is highest among the liquids. Water is used as the standard for a calorie, the calorie is the amount of heat required to raise 1 cm3 of water to 1°C between 15.5 and 16.5°C.
Hence water has a specific heat or heat capacity of 1 cal g-1 degree-1 (1 calorie = 4,184 joules). This high specific heat has importance in terms of rates of heating and cooling of aqueous bodies.
The rate of heating and cooling of plant tissue is governed partly by the heat capacity of water. After the understanding the structure and properties of water molecules it would be appropriate if we move on to the association of water with life.
Water is associated with other bio-molecules to give rise to three different states which includes Solutions, Colloidal (colloidal system) and Suspensions. These three states can be differentiated on the basis of the particle size associated with the water to produce the mixture.
The mixture is a Solution when particle size is very small (particles may be atoms, ions, or molecules) – generally less than 5×10-7 cm (50°A) in diameter.
In this state mostly different ions (H+, OH-, SO4, NO3 etc.) or small molecules (Glucose, Sucrose, etc.) develop association with water.
In contrast a Suspension is a heterogeneous mixture when the particles’ size is usually 10-4 cm or larger in diameter (heavy proteins, hormones, starch etc.).
Whereas the homogeneous distribution of particles between solution and suspension size (light proteins vitamins etc.) is called Colloids.
All these different types of ions or molecules have a definite role to play in the life of the cell. They are either on their way to taking part in some metabolic reaction or they may be the end product of some reaction.
They are vital for life and must be supplied to the cell or be thrown out of the cell promptly and efficiently.
The only means of supply and discharge is their association with water, which delivers them properly with a respectable speed at the place of job.